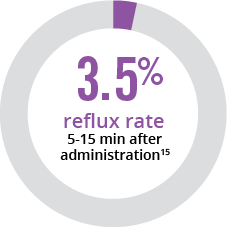
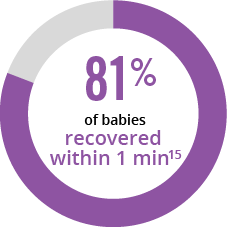
References: 1. CUROSURF® (poractant alfa) Intratracheal Suspension Prescribing Information, Chiesi USA, Inc., May 2021. 2. Moya FR, Gadzinowski J, Bancalari E, et al. Pediatrics. 2005;115:1018-1029. 3. Sinha SK, Lacaze-Masmonteil T, Valls i Soler A, et al. Pediatrics. 2005;115:1030-1038. 4. Survanta® (beractant) Intratracheal Suspension Prescribing Information, AbbVie Inc, October 2020. 5. Infasurf® (calfactant) Intratracheal Suspension Prescribing Information, ONY, Inc, March 2018. 6. Dizdar EA, Sari FN, Aydemir C, et al. Am J Perinatol. 2012;29:95-100. 7. Sandri F, Plavka R, Ancora G, et al. Pediatrics. 2010;125:1402-1409. 8. Dani C, Bertini G, Pezzati M, Cecchi A, Caviglioli C, Rubaltelli FF. Pediatrics. 2004;113:e560-e563. 9. Ramanathan R, Rasmussen MR, Gerstmann DR, Finer N, Sekar K; and the North American Study Group. Am J Perinatol. 2004;21:109-119. 10. Verder H, Albertson P, Ebbesen F, et al. Pediatrics. 1999;103:1-6. 11. Speer CP, Gefeller O, Groneck P, et al. Arch Dis Child Fetal Neonatal. 1995;72:F8-F13. 12. Wiseman IR, Bryson HM. Drugs. 1994;48:386-403. 13. WAC Pricing. First Data Bank, Inc. Accessed September 30, 2024. 14. Karadag N, Dilli D, Zenciroglu A, Aydin B, Beken S, Okumus N. Am J Perinatol. 2014;31:1015-1022. 15. Gerdes JS, Seiberlich W, Sivieri EM, et al. J Pediatr Pharmacol Ther. 2006;11:92-100.