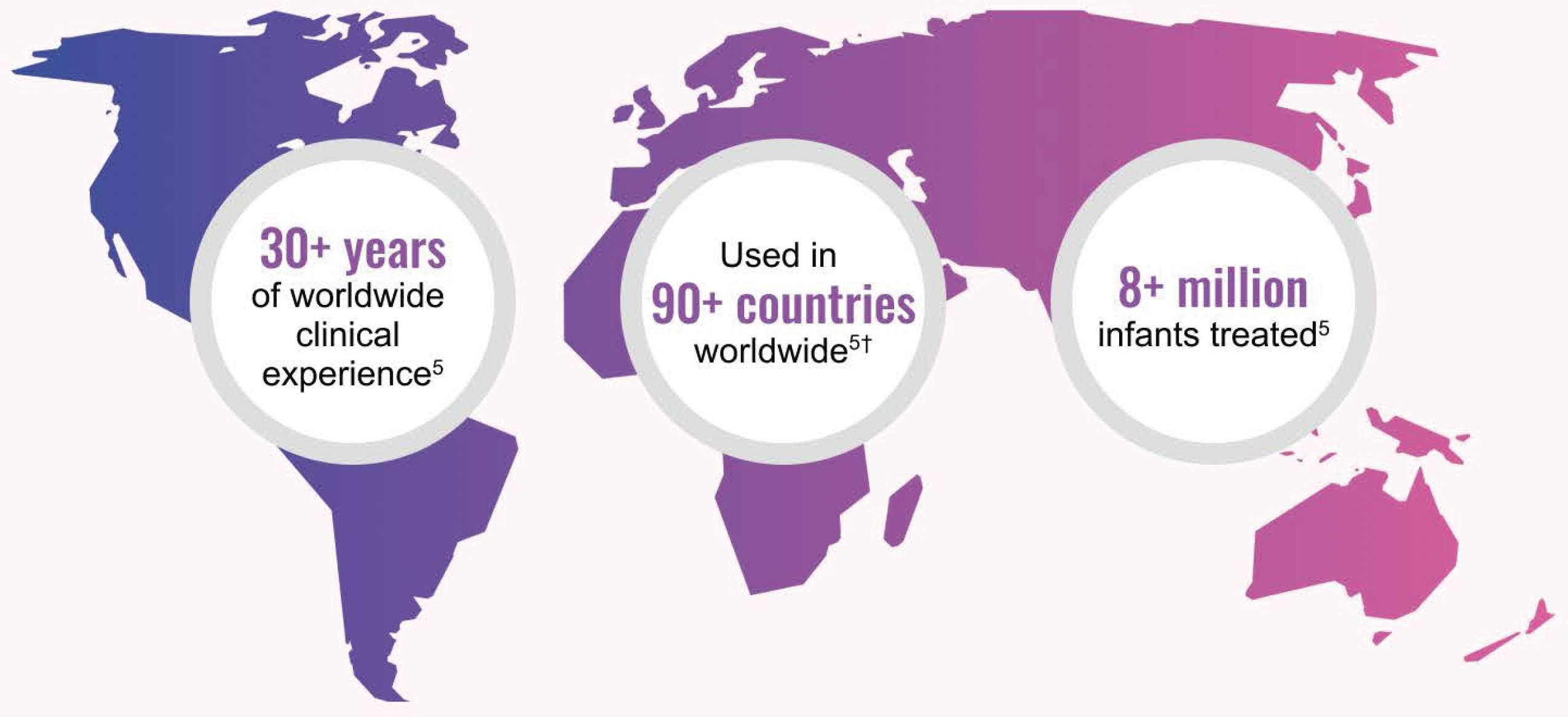
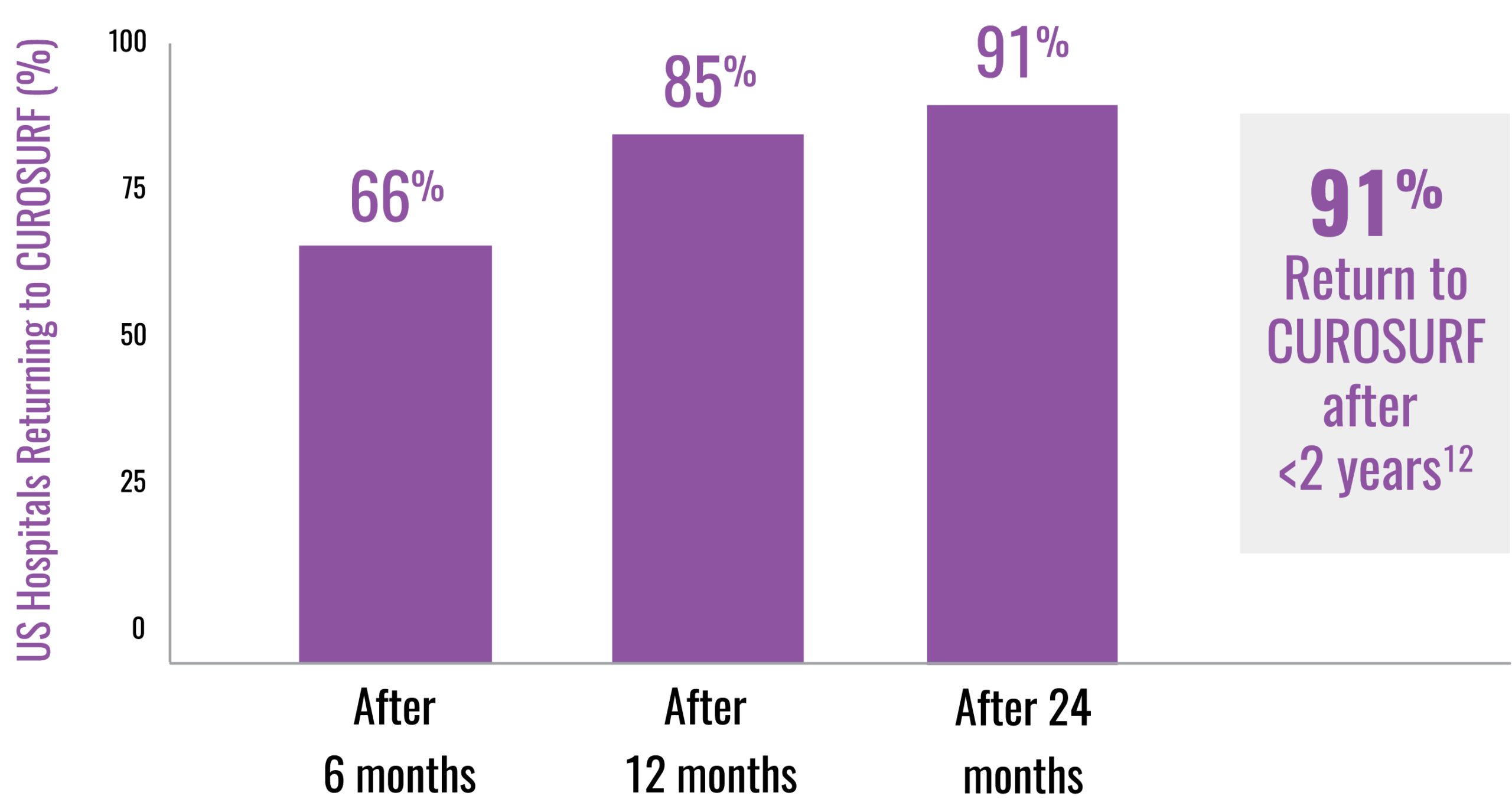
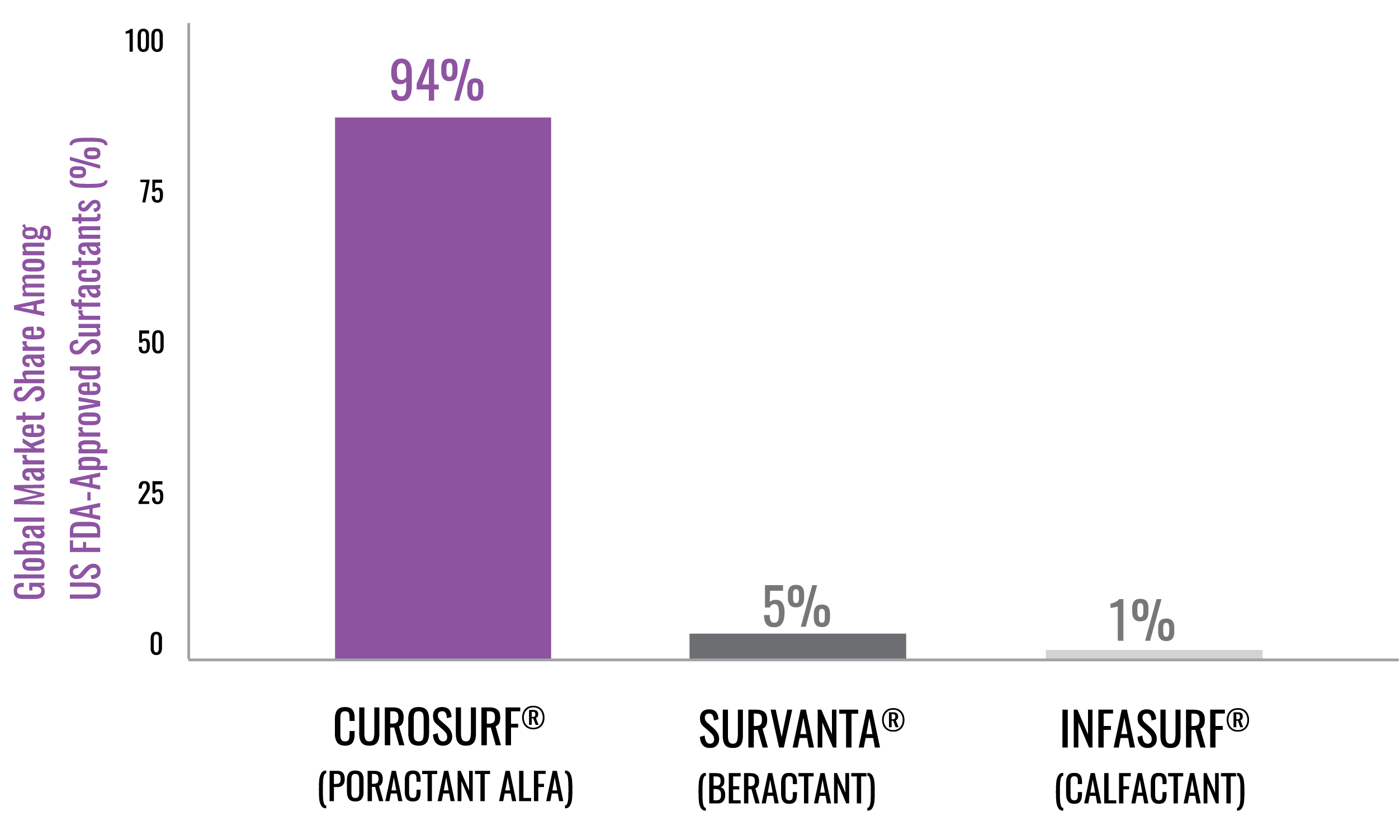References: 1. CUROSURF® (poractant alfa) Intratracheal Suspension Prescribing Information, Chiesi USA, Inc. May 2021. 2. IQVIA Drug Distribution Data, Total Year Q2 2025. 3. IQVIA SMART-US Edition, National Sales Perspectives, All Channels September 2020. 4. IQVIA Global Market Share. Total Year Q2 2025. 5. Data on file, Chiesi Farmaceutici S.p.A. and Chiesi USA, Inc. 6. ONY Biotech website. Accessed January 22, 2025. https://onybiotech.com/products/infasurf 7. US News and World Report Best Children’s Hospitals. Accessed November 14, 2025. https://health.usnews.com/best-hospitals/pediatric-rankings 8. Children’s Hospital Association website. Accessed December 14, 2025. https://www.childrenshospitals.org/hospital-directory. 9. Vermont Oxford Network website. Accessed December 14, 2025. https://public.vtoxford.org/member-map/ 10. US News and World Report Best Hospital for Neonatology. Accessed November 20, 2025. https://health.usnews.com/best-hospitals/pediatric-rankings/neonatal-care 11. Neonatology Fellowship Programs—Neonatology Solutions. Accessed December 24, 2025. 12. Data on file, Chiesi USA, Inc. 13. Survanta (beractant) Intratracheal Suspension Prescribing Information, AbbVie Inc. October 2020. 14. Infasurf (calfactant) Intratracheal Suspension Prescribing Information, ONY, Inc. August 2024. 15. Taeusch HW, Lu K, Ramierez-Schrampp D. Acta Pharmacol Sin. 2002;23(suppl):11-15. 16. Ramanathan R, Rasmussen MR, Gerstmann DR, Finer N, Sekar K; North American Study Group. Am J Perinatol. 2004;21:100-110. 17. Dizdar EA, Sari FN, Aydemir C, Oguz SS, Erdeve O, Uras N, et al. Am J Perinatol. 2012;29:95-100. 18. Gerdes J, Seiberlich W, Sivieri EM, Marsh W, Varner DL, Turck CJ, et al. J Pediatr Pharmacol Ther. 2006;11:92-100. 19. Speer CP, Gefeller O, Groneck P, et al. Arch Dis Child. 1995;72:F8-F13. 20. Collaborative European Multicenter Study Group. Pediatrics. 1988;82:683-691. 21. Verder H, Albertsen P, Ebbesen F, et al. Pediatrics. 1999;103:1-6. 22. Dani C, Bertini G, Pezzati M, Cecchi A, Caviglioli C, Rubaltelli FF. Pediatrics. 2004;113:560-565.